![geometric isomers of [PtBrClNH3py]](../gifs/ccfig9.gif)
3 geometric isomers of a square planar complex [PtBrClNH3(pyr)].
Two or more different compounds having the same formula are called isomers. Two principal types of isomerism are known among coordination compounds. Each of which can be further subdivided.
Ignoring special cases involving esoteric ligands, then:
Geometric isomers are possible for both square
planar and octahedral complexes, but not tetrahedral.
Optical isomers are possible for both tetrahedral and octahedral complexes, but not square planar.
The earliest examples of stereoisomerism involve complexes of Co(III). In 1889, Jorgensen observed purple and green salts of [CoCl2(en)2]+, which Werner later correctly identified as the cis- and trans- geometric isomers. In 1911, the first resolution of optical isomers was reported by Werner and King for the complexes cis-[CoX(NH3)(en)2]2+, where X=Cl- or Br-.
Compound type No. of isomershere a, b, c, and d refer to monodentate ligands.
Ma2b2 2 (cis- and trans-)
Mabcd 3 (use cis- and trans- relations)
A number of examples of these types have been isolated and characterised and they show very different chemical and biological properties. Thus for example, cis-PtCl2(NH3)2 is an anti-cancer agent (cisplatin) whereas the trans- isomer is inactive against cancer (it is toxic), and so not useful in Chemotherapy.
|
|
|
a
|
|
M ----b a----M----b
cis- trans-
![geometric isomers of [PtBrClNH3py]](../gifs/ccfig9.gif)
3 geometric isomers of a square planar complex [PtBrClNH3(pyr)].
Question. Does
cis-amminebromo-cis-chloropyridineplatinum(II)
uniquely identify isomer (ii) above??
Compound type No. of isomershere a, and b, represent monodentate ligands and AA is a bidentate ligand.
Ma4b2 2 (cis- and trans-)
Ma3b3 2 (fac- and mer-)
MAA2b2 3 (2*cis- and 1 trans-)
In the second example, new labels are introduced to reflect the relative positions of the ligands around the octahedral structure. Thus; placing the 3 groups on one face of the octahedral gives rise to the facial isomer and placing the 3 groups around the centre gives rise to the meridional isomer.
The cis- isomer of MAA2b2 may
also exhibit optical isomerism although we will
concentrate largely on optical isomers of the type
M(AA)3 (see below).
Optical isomers are related as non-superimposable mirror images and differ in the direction with which they rotate plane-polarised light. These isomers are referred to as enantiomers or enantiomorphs of each other and their non-superimposable structures are described as being asymmetric.
Various methods have been used to denote the absolute configuration of optical isomers such as R or S, Λ or Δ or C and A. The IUPAC rules suggest that for general octahedral complexes C/A scheme is convenient to use and that for bis and tris bidentate complexes the absolute configuration be designated Lambda Λ (left-handed) and Delta Δ (right-handed).
Priorities are assigned for mononuclear coordination systems based on the standard sequence rules developed for enantiomeric carbon compounds by Cahn, Ingold and Prelog (CIP rules). These rules use the coordinating atom to arrange the ligands into a priority order such that the highest atomic number gives the highest priority number (smallest CIP number). For example the hypothetical complex [Co Cl Br I NH3 NO2 SCN]2- would assign the I- as 1, Br as 2, Cl as 3, SCN as 4, NO2 as 5 and NH3 as 6.
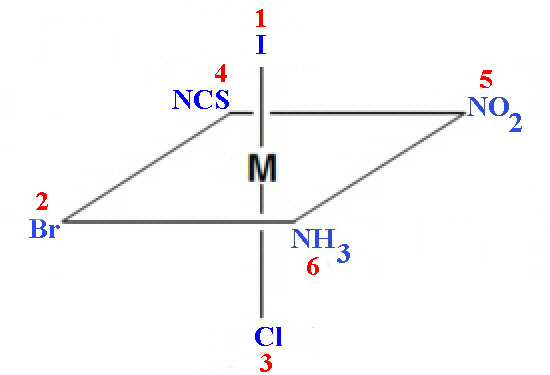
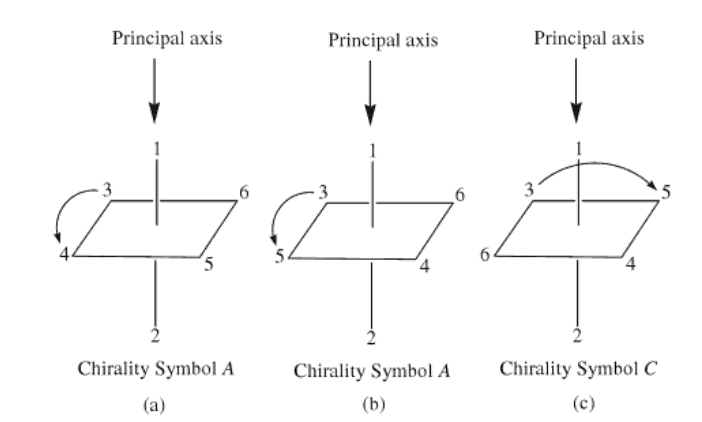
The reference axis for an octahedral centre is that axis containing the
ligating atom of CIP priority 1 and the trans ligating atom of lowest possible
priority (highest numerical value). The atoms in the coordination plane
perpendicular to the reference axis are viewed from the ligand having that
highest priority (CIP priority 1) and the clockwise and anticlockwise sequences
of priority numbers are compared. The structure is assigned the symbol C or A,
according to whether the clockwise (C) or anticlockwise (A) sequence is lower
at the first point of difference. In the example shown above this would be C.
The two optical isomers of [Co(en)3]3+ have identical chemical properties and just denoting their absolute configuration does NOT give any information regarding the direction in which they rotate plane-polarised light. This can ONLY be determined from measurement and then the isomers are further distinguished by using the prefixes (-) and (+) depending on whether they rotate left or right.
3_isomers.jpg)
|
left-handed Λ isomer |
and right-handed Δ isomer |
To add to the confusion, when measured at the sodium D line
(589nm), the tris(1,2-diaminoethane)M(III) complexes (M= Rh(III)
and Co(III)) with IDENTICAL absolute
configuration, rotate plane polarised light in
OPPOSITE directions!
The left-handed (Λ)-[Co(en)3]3+
isomer gives a rotation to the right and therefore corresponds to
the (+) isomer.
Since the successful resolution of an entirely inorganic ion (containing no C atoms) (hexol) only a handful of truly inorganic complexes have been isolated as their optical isomers e.g. (NH4)2Pt(S5)3.2H2O.
For tetrahedral complexes, R and S would be used in a similar method to tetrahedral Carbon species and although it is predicted that tetrahedral complexes with 4 different ligands should be able to give rise to optical isomers, in general they are too labile and can not be isolated.
one isomer [Co(NH3)6] [Cr(C2O4)3]
another isomer [Co(C2O4)3] [Cr(NH3)6]
Notice that both anions are necessary to balance the charge of the complex, and that they differ in that one ion is directly attached to the central metal but the other is not. A very similar type of isomerism results from replacement of a coordinated group by a solvent molecule (Solvate Isomerism). In the case of water, this is called Hydrate isomerism.one isomer [PtBr(NH3)3]NO2 -> NO2- anions in solution
another isomer [Pt(NO2)(NH3)3]Br -> Br- anions in solution
These isomers have very different chemical properties and on reaction with AgNO3 to test for Cl- ions, would find 1, 2, and 3 Cl- ions in solution respectively.[CrCl2(H2O)4]Cl.2H2O bright-green
[CrCl(H2O)5]Cl2.H2O grey-green
[Cr(H2O)6]Cl3 violet
[Co(ONO)(NH3)5]Cl the nitrito isomer -O attached
[Co(NO2)(NH3)5]Cl the nitro isomer - N attached.
l. Dyes and Pigments: Coordination compounds have been used from the earliest times as dyes and pigments, for example madder dye which is red, was used by the ancient Greeks and others. It is a complex of Hydroxyanthraquinone. A more modern example is the pigment copper phthalocyanine, which is blue.
2. Analytical Chemistry: You have already encountered many such uses during the laboratory course.
3. Sequestering Agents: Related to their use as masking agents is the use of ligands for "sequestering" i.e. for the effective removal of objectionable ions from solution in industrial processing, e.g. EDTA is used to "soften" water. The addition of EDTA to water is used in boilers etc., to prevent "scaling" or build up of insoluble calcium salts.
4. Extraction of Metals: Sometimes certain metals can be leached from their ores by formation of stable complexes e.g. Ag and Au as complexes of cyanide ion.
5. Bio-Inorganic Chemistry: Naturally
occurring complexes include haemoglobin, chlorophyll, vitamin B12
etc.
EDTA and other complexing agents have been used to speed the
elimination of harmful radioactive and other toxic elements from
the body. (e.g. Pb2+). In these cases soluble metal
chelate complexes are formed.
6. Chemo-therapy: an example here is the use of cis-PtCl2(NH3)2 as an anti-tumour drug.
 Return to Chemistry, UWI-Mona,
Home Page
Return to Chemistry, UWI-Mona,
Home Page